Possible Treatments For Stargardt's Disease
Currently, there is no known treatment for this disease. However, in order to slow the progression of Stargardt's disease, people with Stargardt’s disease are often recommended to protect their eyes from bright sunlight by wearing sunglasses and hats. In addition to this, they are also advised not to eat foods that are high in vitamin A or use vitamin A supplements or beta-carotene supplements. This is very important in that a toxic build up of vitamin A is what is constantly killing the photoreceptors of the eye. By not putting much extra vitamin A in the body, one can make sure that they are not contributing to the build up of vitamin A, the thing that is killing their photoreceptor cells. These are very easy things to do that could help slow the progression of Stargardt's Disease.
The use of stem cells could have an enormous impact on the treatment of Stargardt's disease. The only treatment given to people with Stargardt's disease currently and for many years was advice to stay out of the sun and keep vitamin A out of their diet. However, treatments such as ones involving stem cells have been gaining discussion and research in recent years. These treatments can potentially temporarily restore vision or even cure the disease altogether.
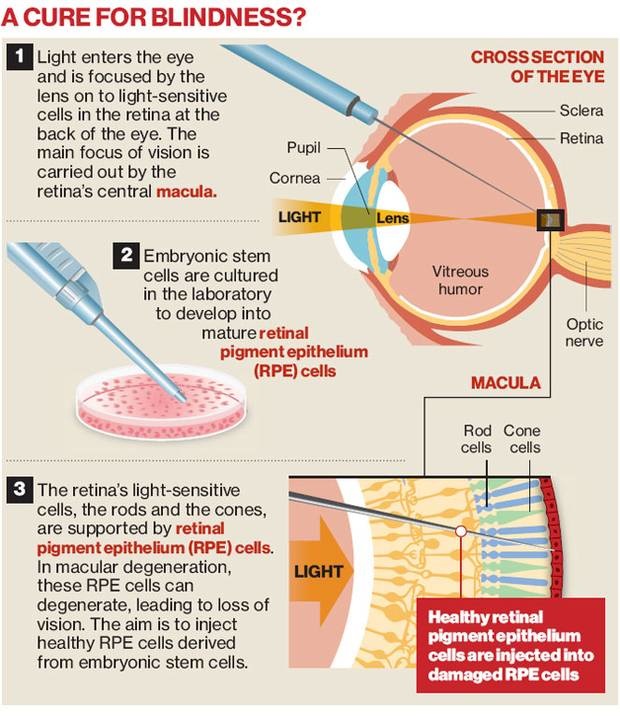
Stem cells could repair age related macular degeneration, but not macular degeneration caused by genetic defects, such as what occurs in inherited genetic diseases, like Stargardt's disease. In the case of Stargardt's disease, the photoreceptor cells of the retina could not be treated or cured of Stargardt's disease as the problem does not lie within the photoreceptor cells, within a faulty gene in a person's DNA. The photoreceptor cells do not die on their own; they die due to the fact that not all of the processes in the retina are not working properly which causes the retina to be unable to clear toxic A2E out of itself, causing the the photoreceptor cells to die.
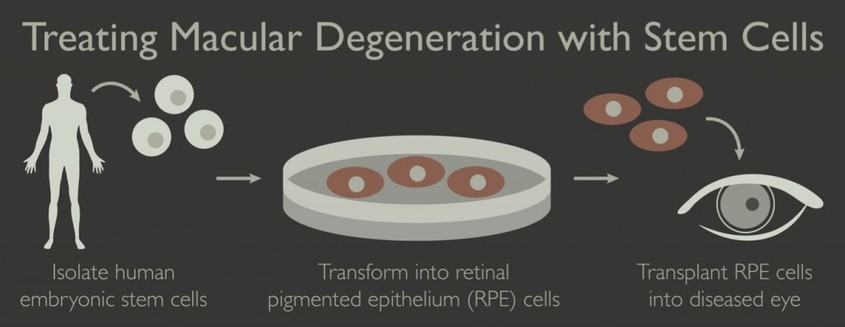
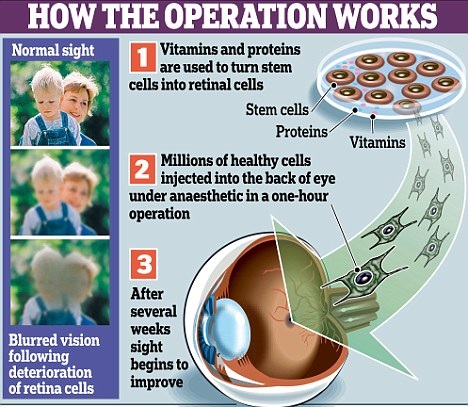
However, a solution can be found to almost any problem. Scientists can create stem cells out of a sliver of skin from the arm of the person they are treating. They could then use a mix of vitamins and proteins to coax the stem cells into growing into retinas. The photoreceptor cells could then be extracted from those retinas and inserted into the retina of a person with Stargardt's disease, temporarily restoring the person's vision by giving them new, working photoreceptor cells to use to absorb light to convert into signals that can stimulate biological processes. Currently, scientist have not been able to get the human retina to fully mature, but to only mature to the point at which a baby's retina would be before it is born. However, with more time and research, scientist think that it is very possible to get the retinas to mature fully before inserting the photoreceptors cells into a patient's eye through a one hour surgery. In addition to replacing dead photoreceptor cells, stem cells grown into photoreceptor cells early on can create chemicals that can save the retina by slowing the degeneration of all ready existing photoreceptor cells. These chemicals are known as growth factors. Although this would work well for repairing the sight of a person with age related macular degeneration, this would not work well for a person with Stargardt's Disease as the degeneration of cells is not just coming from wear-and-tear over time, but of the constant build up toxic material in the eye caused by the occurrence of a faulty gene. Despite this restoration in vision that the insertion of new photoreceptor cells causes, eventually these photoreceptor cells would die as well once too much A2E builds up in the eye again, causing the person's vision to worsen back to the state it was once at if no more photoreceptor cells were added to the retina. By adding new photoreceptor cells back into the retina, a person with Stargardt's disease could have their vision restored temporarily, but not have their disease cured. To cure Stargardt's disease, the defective gene that causes the disease would have to be fixed so that the processes in the eye occur as they are supposed to in order to prevent the toxic A2E from building up. As of right now, it seems that this might be the best way to restore vision in patients. Although it will not last forever, it could restore vision a significant amount for several years in a more minimally invasive way. It is also more known how this will work than the the other treatments at this point.
The use of stem cells could have an enormous impact on the treatment of Stargardt's disease. The only treatment given to people with Stargardt's disease currently and for many years was advice to stay out of the sun and keep vitamin A out of their diet. However, treatments such as ones involving stem cells have been gaining discussion and research in recent years. These treatments can potentially temporarily restore vision or even cure the disease altogether.
Stem cells could repair age related macular degeneration, but not macular degeneration caused by genetic defects, such as what occurs in inherited genetic diseases, like Stargardt's disease. In the case of Stargardt's disease, the photoreceptor cells of the retina could not be treated or cured of Stargardt's disease as the problem does not lie within the photoreceptor cells, within a faulty gene in a person's DNA. The photoreceptor cells do not die on their own; they die due to the fact that not all of the processes in the retina are not working properly which causes the retina to be unable to clear toxic A2E out of itself, causing the the photoreceptor cells to die.
However, a solution can be found to almost any problem. Scientists can create stem cells out of a sliver of skin from the arm of the person they are treating. They could then use a mix of vitamins and proteins to coax the stem cells into growing into retinas. The photoreceptor cells could then be extracted from those retinas and inserted into the retina of a person with Stargardt's disease, temporarily restoring the person's vision by giving them new, working photoreceptor cells to use to absorb light to convert into signals that can stimulate biological processes. Currently, scientist have not been able to get the human retina to fully mature, but to only mature to the point at which a baby's retina would be before it is born. However, with more time and research, scientist think that it is very possible to get the retinas to mature fully before inserting the photoreceptors cells into a patient's eye through a one hour surgery. In addition to replacing dead photoreceptor cells, stem cells grown into photoreceptor cells early on can create chemicals that can save the retina by slowing the degeneration of all ready existing photoreceptor cells. These chemicals are known as growth factors. Although this would work well for repairing the sight of a person with age related macular degeneration, this would not work well for a person with Stargardt's Disease as the degeneration of cells is not just coming from wear-and-tear over time, but of the constant build up toxic material in the eye caused by the occurrence of a faulty gene. Despite this restoration in vision that the insertion of new photoreceptor cells causes, eventually these photoreceptor cells would die as well once too much A2E builds up in the eye again, causing the person's vision to worsen back to the state it was once at if no more photoreceptor cells were added to the retina. By adding new photoreceptor cells back into the retina, a person with Stargardt's disease could have their vision restored temporarily, but not have their disease cured. To cure Stargardt's disease, the defective gene that causes the disease would have to be fixed so that the processes in the eye occur as they are supposed to in order to prevent the toxic A2E from building up. As of right now, it seems that this might be the best way to restore vision in patients. Although it will not last forever, it could restore vision a significant amount for several years in a more minimally invasive way. It is also more known how this will work than the the other treatments at this point.
Not only can photoreceptor cells be replaced, retinal pigmented epithelial (RPE) can be replaced as well in order to restore people's vision. RPE cells provide essential supportive, functions for photoreceptors. By placing healthy RPE cells into the retina, researchers think that they can save photoreceptor cells and slow or halt vision loss. This is also a treatment that is well suited to treating this disease. Like the photoreceptor cell placement, not only is this treatment less invasive, but there is more known about it will work than other treatments.
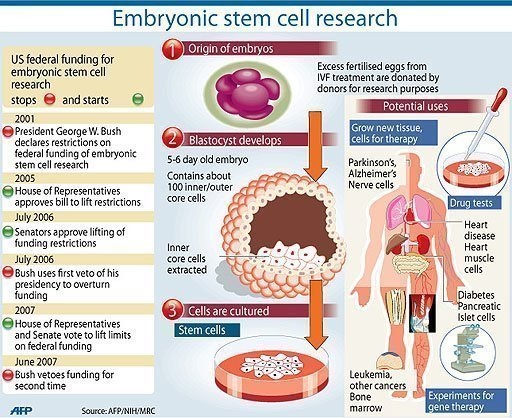
Genetic engineering combined with stem cells for the purpose of gene therapy is another hypothetical way that Stargardt's Disease could be treated or cured. Stem cells could be created and manipulated to grow into retinas in the process previously mentioned. While these stem cells are still growing into retinas and are still replicating, genetic engineering could then be used to correct the mutation within their DNA. This could be done by forming recombinant DNA out of a plasma and a section of DNA containing a working ABCA4 gene. A restriction enzyme would cut the needed sections of DNA out in order to gather the materials needed to make this recombinant DNA with a functional ABCA4 gene. These two new pieces of DNA could then be connected by their sticky ends and inserted into the still forming and replicating retinal cells.
The new replicating cells would all have a working ABCA4 gene. These new photoreceptor cells could then be placed into the retina. Because these cells would have a working ABCA4 gene, the phototransduction cycle would be able to complete itself. This would cause lipofuscin (essentially vitamin A) to be cleared out of the eye regularly, preventing any new cells from dying. With this method, it is possible that the eyes could then be exactly the same as they were before any new cells were inserted into them, except now they would have a working ABCA4 gene, therefore curing that person's Stargardt's disease, restoring their vision, and preventing any more photoreceptor cells from dying.
Genetic engineering combined with stem cells for the purpose of gene therapy is another hypothetical way that Stargardt's Disease could be treated or cured. Stem cells could be created and manipulated to grow into retinas in the process previously mentioned. While these stem cells are still growing into retinas and are still replicating, genetic engineering could then be used to correct the mutation within their DNA. This could be done by forming recombinant DNA out of a plasma and a section of DNA containing a working ABCA4 gene. A restriction enzyme would cut the needed sections of DNA out in order to gather the materials needed to make this recombinant DNA with a functional ABCA4 gene. These two new pieces of DNA could then be connected by their sticky ends and inserted into the still forming and replicating retinal cells.
The new replicating cells would all have a working ABCA4 gene. These new photoreceptor cells could then be placed into the retina. Because these cells would have a working ABCA4 gene, the phototransduction cycle would be able to complete itself. This would cause lipofuscin (essentially vitamin A) to be cleared out of the eye regularly, preventing any new cells from dying. With this method, it is possible that the eyes could then be exactly the same as they were before any new cells were inserted into them, except now they would have a working ABCA4 gene, therefore curing that person's Stargardt's disease, restoring their vision, and preventing any more photoreceptor cells from dying.
There is another hypothetical way of curing Stargardt's Disease: the cloning of an embryo combined with the usage of gene therapy to fix the defective ABCA4 gene in the stem cells from this embryo to insert back into the mother's womb could potentially cure Stargardt's disease.
If a couple knows that they are carriers for the recessive gene that causes Stargardt’s disease and they are planning on having children and are afraid that their child will inherit the disease, the couple can first produce an embryo through in vitro fertilization. The embryo could then be screened of the genetic abnormality. If the embryo had the genetic abnormality, stem cells would be taken from the embryo when it is six day old. Scientist would then use genetic engineering techniques to correct the genetic abnormality. The corrected stem cell nucleus would then be inserted into an egg to form a new embryo that would then be implanted into the mother's womb. This would cause the embryo to grow into a fetus that would essentially be an identical twin of the original embryo but with the abnormal gene corrected in every one of its cells. This fetus would still be a clone, but of an entirely new individual who is a combination of both parents. The clone would be a child of both parents, like any other child, not a clone of one parent. This child would not have Stargardt's disease as a result of a combination of cloning and genetic therapy. This method would only work to cure Stargardt's disease in embryos that will later grow into adults. There is no known cure for Stargardt's disease that has been diagnosed in people who are already young children or adults. Currently this might only work with embryos, fetuses, and much less likely, infants. Although this method may be controversial or it may not have been practiced yet, this is likely a nearly definite and reliable way of curing this disease that is also fairly minimally invasive. All of the steps in this process have been done before; they just have never been brought together in quite this way before. Because all of the steps in this process have been done before (scientist have clone animals, fixed genetic mutations through gene therapy, implanted a fertilized egg in a womb) and some of the steps have even been brought together before, this way of curing this disease seems more reliable and less invasive or harmful than other methods.
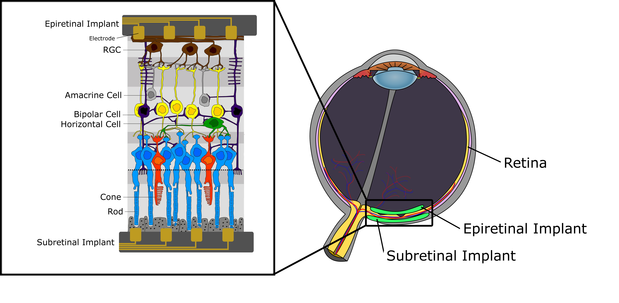
Another way of restoring vision is through retinal implants. A retinal implant is a biomedical implant technology currently being developed by a number of private companies and research institutions worldwide. The implant is meant to partially restore useful vision to people who have lost their vision due to degenerative eye conditions. There are three types of retinal implants currently in clinical trials: epiretinal implants which go on the retina, subretinal implants which go behind the retina, and suprachoroidal implants, which go above the vascular choroid. Retinal implants provide the patient who gets them with low resolution images by electrically stimulating surviving retinal cells. Such images may restore specific visual abilities, such as light perception and object recognition.
In order to carry out any of this treatment that involves the changing of an organism's DNA or cells, different types of biotechnology are going to have to be used on order to make this happen. It would be impossible to do these things without it. Biotechnology is extremely important for this reason. Biotechnology allows humans to alter the DNA of a person or to repair damage that has been done to the body. It allows humans to see extremely small things, or to see within the human body. It has allowed humans to perform complex surgeries in minimally invasive ways and extend their senses beyond what they ever could sense naturally. For these reasons, biotechnology is incredibly important. It has allowed people to live longer, better quality lives.
There are many different types of technology that are being used to help people with Stargardt's disease continue to be able to see the world around them. For example, there are many different types of visual aids that will magnify the words in a book or on a page so that people with Stargardt's can continue to be able to read and write before they lose too much of their central vision to do so. There is also another piece of technology that is becoming increasingly popular that is helping people with low vision see the world around them. This technology is known as eSight. eSight almost looks like a large pair of goggles that a person will wear over their eyes whilst holding a controller. Enhanced video is transmitted to the "goggles" from a controller and displayed on LED screens in front of the wearer's eyes. The controller can be used to zoom in on details up to 14 times their original size, which means that wearers can see a far-away object such as a clock on a wall or read a newspaper. The controller uses different algorithms and selected settings that customizes the video for the person so that they can see. The technology helps people who have some low vision see people’s faces, read newspaper and signposts, use computers and watch TV, for example, because the glasses can automatically focus on short, medium and long-range objects.
eSight, biotechnology that allows a person with low vision to see much more clearly.
http://sitn.hms.harvard.edu/wp-content/uploads/2014/01/Lamkin-Figure-1024x397.jpg
http://i.dailymail.co.uk/i/pix/2011/09/23/article-2040789-0E0AD53B00000578-500_468x407.jpg
http://medblog.medlink-uk.net/bilalmohammed/files/2012/04/pg-25-blindness-graphic.jpg
http://churchandstate.org.uk/wordpressRM/wp-content/uploads/2010/11/hescr1.jpg
http://www.crunchwear.com/wp-content/uploads/2013/08/eyewear-image.jpg
http://i.dailymail.co.uk/i/pix/2011/09/23/article-2040789-0E0AD53B00000578-500_468x407.jpg
http://medblog.medlink-uk.net/bilalmohammed/files/2012/04/pg-25-blindness-graphic.jpg
http://churchandstate.org.uk/wordpressRM/wp-content/uploads/2010/11/hescr1.jpg
http://www.crunchwear.com/wp-content/uploads/2013/08/eyewear-image.jpg